Meet The Interns – Summer 2017
Summer is an exciting time for us here at Auxo Medical because we get to welcome a new group of incredible people to the family. We’re so excited to introduce the newest members of the Auxo team, the Summer 2017 interns!
These three students are working hard and are determined to make this the best summer yet at Auxo Medical. We’ve asked the interns to answer a few questions in order to get to know them a little more. Without further ado, please welcome the Summer 2o17 Interns!



 So says the Occupational Safety and Health Administration (OSHA). One study put the incidence rate for work related nonfatal injuries and illnesses in health care and social assistance at nearly 40% higher than that experienced in all private industry.
So says the Occupational Safety and Health Administration (OSHA). One study put the incidence rate for work related nonfatal injuries and illnesses in health care and social assistance at nearly 40% higher than that experienced in all private industry.
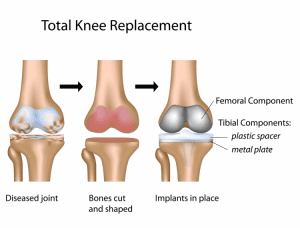

 Features:
Features:


 Here’s a reminder of the recent ban of powdered gloves by the Federal Drug Administration (FDA). Compliance is mandatory in less than a week.
Here’s a reminder of the recent ban of powdered gloves by the Federal Drug Administration (FDA). Compliance is mandatory in less than a week.